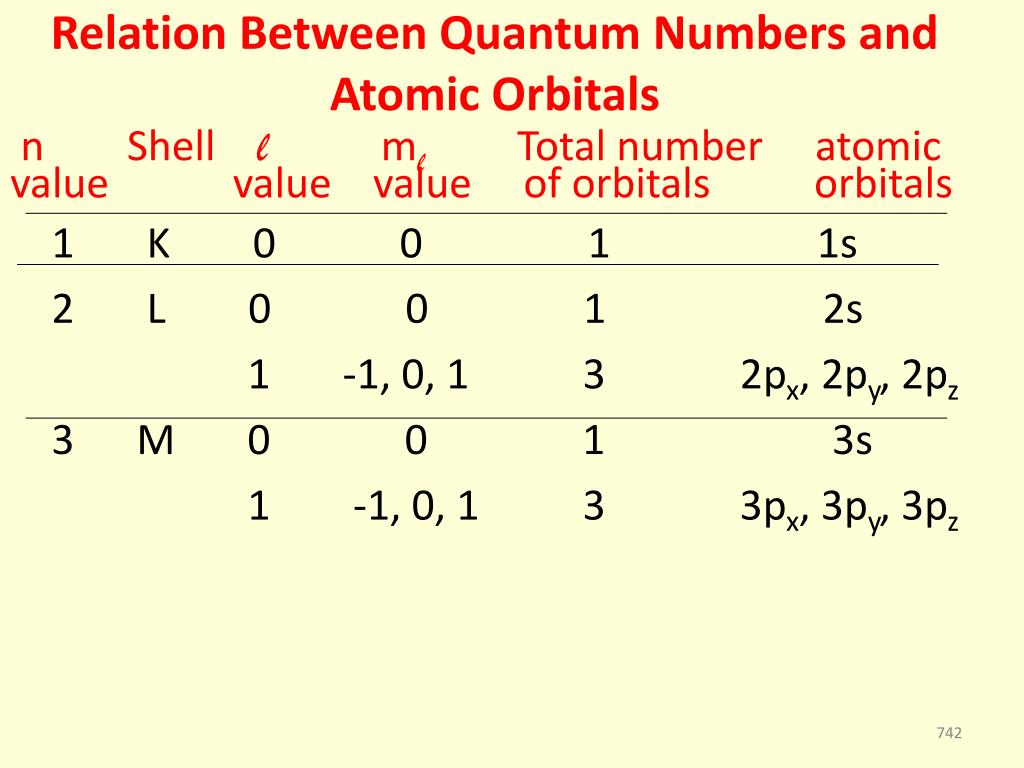
#=> (n, l, m_l, m_s) = (4, 1,, -1/2) =># represents only one of 35 electrons in the neutral Bromine atom. Give a possible set of all four quantum numbers for a 4p electron of Rb. The 4 Quantum Numbers for the 'last' electron to fill a Bromine electronic configuration would be the electron in the half-filled #4p# orbital. The principal quantum number species the number of shells, the azimuthal quantum number specifies the shape of orbital, magnetic quantum number tells about the orientation of orbital and spin quantum number species the spin of electron. Spin QN ( #m_s#) => the spin of the electron, up or down #pm 1/2#, respectively).Magnetic QN ( #m_l#) => Orientation of orbital in 3 dimensions.Orbital QN ( #l#) also known as Angular Momentum QN => Shape of Orbital.Principle QN ( #n#) => Defines Principal Energy Level.The states are represented by symbols, and special. Thus, for n 1, there is only one state (1,0,0). Each set of quantum numbers is called a state. For n 1, the only possible value for quantum number l is 0, and m 0. The 4 Quantum Numbers (QN) and what they describe are: follow the rules: Rules are algorithms, by which we generate possible quantum numbers. Think of the set of Quantum Numbers for an electron as being a 'discrete energy window' for that electron and no other electron in the element's electron configuration can occupy the same energy window. 
This is significant in that elements with multiple electrons cannot have more than two in a single orbital. So, you know that you must find a valid combination of quantum numbers that can describe the position and spin of an electron located in a 5p-orbital. 4 Quantum Numbers define the character of an electron in an energy specific orbital.Īccording to the Pauli Exclusion principle, no two electrons can have the same set of 4 quantum numbers. As you know, four quantum numbers are used to describe the position and spin of an electron inside an atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
